 
Some are even capable of upmixing 5.1 channel audio to 7.1, which is excellent if your headphones support 7.1 channels and your audio sources don’t.Ĭonnectivity - Look for a sound card with the jacks you need to plug in your equipment. You generally want a sound card with a signal-to-noise ratio over 100dB, but the best sound cards are in the 124dB range, which is a significant improvement.Ĭhannels - Many decent, budget-friendly sound cards typically support 5.1 channel audio, but you can spend a little more to get one that can handle 7.1 surround sound. 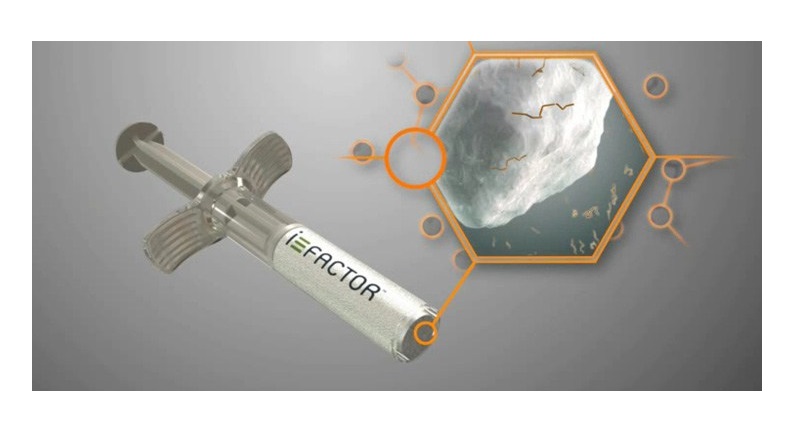
Cerapedics is headquartered in Westminster, CO.Audio quality - The overall audio quality of a sound card is a complicated equation that depends on the signal-to-noise ratio, frequency response, and total harmonic distortion. Cerapedics' next-generation product is currently being evaluated for use in lumbar interbody fusion through ASPIRE, a pivotal clinical study, and has been granted a Breakthrough Device Designation by the FDA. i-FACTOR is a Class III FDA approved product indicated for single-level ACDF. Bone grafts, including Cerapedics' products, are used in over four million annual spine, orthopedics, trauma and interventional procedures worldwide. Cerapedics has hired more than 100 employees in Colorado in the last five years and plans to hire another 60 Colorado-based employees by the end of 2024.Ĭerapedics is a global, commercial-stage orthopedics company that aspires to redefine the standard of care for bone repair by healing bones faster and at higher rates, without compromising safety, so that patients can live their healthiest lives. This next generation product has been granted breakthrough status by FDA and is being studied under an investigational device exemption (IDE).īy expanding its facility, Cerapedics is adding over 50 percent more space to its Colorado headquarters to increase space for manufacturing, research and development and other key functions. local autograft in overall success (defined as meeting Fusion, Function (Neck Disability Index), Neurological and Safety endpoints) at one and two-year endpoints for single-level Anterior Cervical Discectomy and Fusion.Ĭerapedics has fully enrolled its 290-patient randomized controlled clinical study evaluating the safety and efficacy of the next-generation P-15 Peptide Enhanced Bone Graft in transforaminal lumbar interbody fusion (TLIF) surgery. i-FACTOR has proven statistical superiority vs. 
I-FACTOR is the only spinal bone graft powered by P-15 Osteogenic Cell Binding Peptide™, and it is just one of two FDA approved Class III drug-device bone grafts with a spinal indication. "We are fortunate to be based in Colorado and to recruit from the area's experienced, innovation-focused talent pool as we prepare to launch our next-generation product." We are committed to supporting the needs of surgeons and their patients by expanding our facility," said Valeska Schroeder, Chief Executive Officer of Cerapedics. "Since the launch of i-FACTOR, we have seen a continuous increase in its use in spinal fusion. (PRNewsfoto/Cerapedics Inc.) (PRNewswire) This novel mechanism of action is designed to support safer and more predictable bone formation compared to commercially available bone growth factors. i-FACTOR Peptide Enhanced Bone Graft is the only biologic bone graft in orthopedics that incorporates a small peptide as an attachment factor to stimulate the natural bone healing process. Cerapedics is an ortho-biologics company focused on developing and commercializing its proprietary small peptide (P-15) technology platform. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
